Calculating pH, pOH, [H+], [OH-]
Now that you've learned a lot about acids and bases, it's time to learn how to calculate pH, pOH, [H+], and [OH-] of a solution.
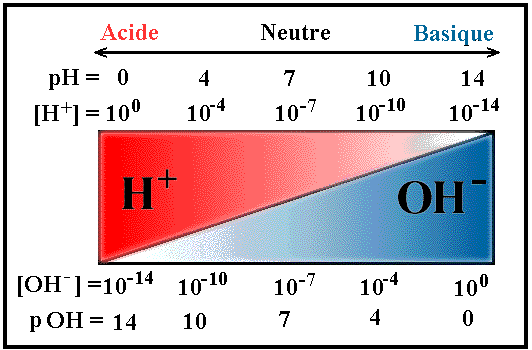
Before you get to the formulas, its important to know more about pH and pOH. You should already know that [H+] is the concentration of hydrogen ions (indicated by the brackets) and that [OH-] is the concentration of hydroxide ions (again, indicated by the brackets).
|
pH
|
pOH
|
Here are the formulas for calculating pH, pOH, [H+], [OH-]:
pH= -log[H+]
[H+]= 2nd log(-pH)
pOH + pH=14
pOH= -log(OH-)
[H+]= 2nd log(-pH)
pOH + pH=14
pOH= -log(OH-)
**Don't know what log is? Log is a function on a graphing calculator and can be found here:
Example Problems:
If a solution has a pH of 3, what is the [H+]?
If a solution has a pH of 3, what is the [H+]?
- Pick one of the formulas: in this case, we are finding [H+], so the formula is: [H+]=2nd log (-pH)
- Plug in the information into the formula: [H+]= 2nd log(-3)
- Enter and look on the graphing calculator for the answer: [H+]= 0.001
- Write the answer in scientific notation if necessary: [H+]= 1 x 10^-3
- Pick one of the formulas: in this case, we are finding pH, so the formula is: pH= -log[H+]
- Plug in the information into the formula: pH=-log[0.2M]
- Enter and look on the graphing calculator for the answer: pH= 0.699
- Pick one of the formulas: in this case, we are finding pOH and pH is known, so the formula is: pOH + pH=14
- Plug in the information into the formula: pOH + 0.699 =14
- Enter and look on the graphing calculator for the answer: pOH= 13.3
If you fully understand how to calculate pH, pOH, [H+], and [OH-], click here to take a quiz on your skills!