Buffers
A buffer is a solution that is resistant to pH when small quantities of an acid or a base are added to it. Buffers consist of a weak conjugate acid-base pair. This means it's either composed of a weak acid and its conjugate base, or a weak base and its conjugate acid. However, this depends on the desired pH.
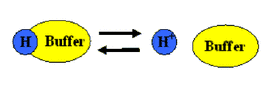
How it Works:
A buffer solution has to contain things which will remove any hydrogen ions or hydroxide ions that you might add to it, otherwise the pH will change. A buffer is able to resist pH change because the conjugate acid and conjugate base are both present in observable amounts and are able to neutralize small amounts of other acids and bases when they are added to the solution.
A buffer solution has to contain things which will remove any hydrogen ions or hydroxide ions that you might add to it, otherwise the pH will change. A buffer is able to resist pH change because the conjugate acid and conjugate base are both present in observable amounts and are able to neutralize small amounts of other acids and bases when they are added to the solution.
|
d
|
Example of a Buffer- Hydrofluoric Acid (HF) and Sodium Fluoride (NaF):
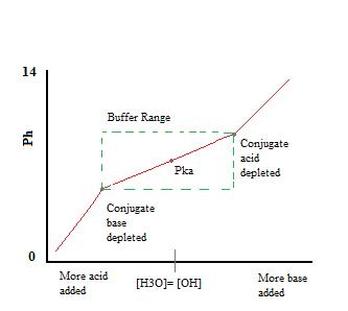
- Hydrofluoric acid (HF) is a weak acid, so it will partially dissociate: HF+H2O → F+H3O - Sodium Fluoride (NaF) is added into the solution and is mixed in until the desired pH volume and pH are reached - When NaF fully dissolves in water, the reaction is: NaF+H2O → Na+F - Na is the conjugate of a strong base, meaning that it will not affect the pH of reactivity of the buffer - The NaF will increase the concentration of F in the solution, leading to less dissociation of HF in the original equation - The amounts of the conjugate acid (HF) and the conjugate base (F) allow the solution to function as a buffer (as seen on the graph on the left) - Based off this graph, it is evident that a larger amount of conjugate acid or base will have a greater buffering capacity |
Real Life Applications of Buffers:
2. Buffers are used in shampoos to balance out the alkalinity that would normally burn your scalp. Citric acid and sodium hydroxide are two examples of buffs used for shampoo
3. Buffers are used in the process of making alcohol, called fermentation. To make sure the solution isn't too acidic, buffers are put in before fermentation.
|
So now that you know about buffers, it's time to move on to strength and concentration by clicking here.