Molarity
|
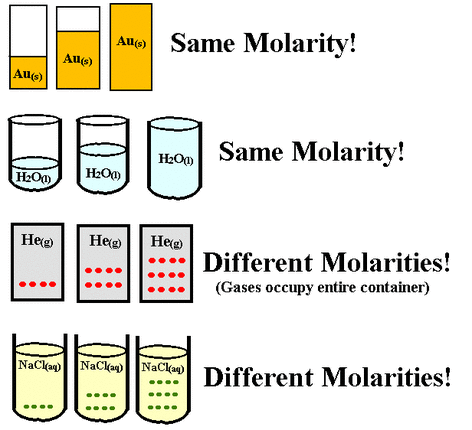
Molarity is…
the number of moles of solute in one liter of solution. Molarity is used... when talking about acids, alkalis, and other solutions. The unit for molarity is… called a molar and is abbreviated as "M". Molarity is measured by… concentration (which is the amount of solute in a solution that is measured in moles). |
THE CHEMICAL EQUATION FOR MOLARITY:
Molarity (M)= Moles (mol)/volume (liters)
Molarity (M)= Moles (mol)/volume (liters)
After learning about molarity, click here to learn about the calculations: pH, pOH, [H+], [OH-]