Titrations
|
Introduction to Titrations:
The Two Forms of Titrations:
|
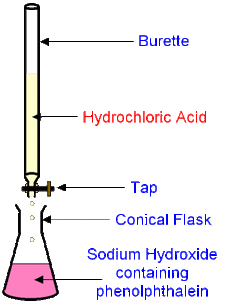
How to Execute a Titration in a Lab:
- Tie hair back, take out goggles, and put on a smock if wanted.
- Take out materials.
- Label a 250 mL beaker A for acid and another 250 mL beaker B for base
- Wash and dry materials thoroughly so there is no contamination.
- Pour any quantity (cc) of hydrochloric acid (HCl) into beaker A
- Pour any quantity (cc) of sodium hydroxide (NaOH) into beaker B
- Clean out the burette with water and then with HCl to prevent dilution
- Attach the burette to the clamp stand and place funnel at the top of the burette.
- Take beaker A and pour at least 10 cc of HCl into the burette through the funnel
- Record the initial amount of HCl in the burette
- Take out a conical flask and wash it with water
- Take out a volumetric pipette and clean it out with water and then with NaOH to prevent dilution
- Using the volumetric pipette, measure exactly 10 cc of NaOH from beaker B
- Release the 10 cc of NaOH from the volumetric pipette into the flask
- Using a pipette, add in three drops of phenolphthalein into the flask with NaOH
- Place the flask with the NaOH below the burette with the HCl
|
To perform a rough titration:
OVERALL GOAL: In this lab, the aim was to neutralize the 10 cc of NaOH in the flask with exactly 10 cc of HCl
|
To perform an exact titration:
|
However, if the molarity of a solution is unknown, use the formula: M1V1 = M2V2
M1= molarity of first solution
V1= volume of first solution
M2= molarity of second solution
V2= volume of second solution
For example…
A titration is completed and you need to find the molarity of 10 cc of HCl. You are mixing this with 10cc of 1 mole of NaOH.
So, using the formula...
M1V1=M2V2
M(10)=1(10)
M=1
M1= molarity of first solution
V1= volume of first solution
M2= molarity of second solution
V2= volume of second solution
For example…
A titration is completed and you need to find the molarity of 10 cc of HCl. You are mixing this with 10cc of 1 mole of NaOH.
So, using the formula...
M1V1=M2V2
M(10)=1(10)
M=1
***To watch an actual titration of HCl and NaOH, click on the video below:

